“Nightmare.”
That’s how one man described a weeks-long ordeal involving ad hoc surgery, repeated infections and some hefty out-of-pocket fees – all caused by a device that was once thought to be a miracle for those dealing with the visible signs of HIV.
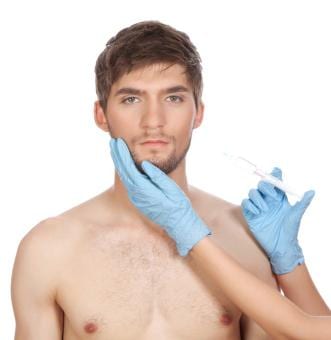
The man was suffering from lipoatrophy – also known as facial wasting – a side effect of the antiretroviral drug cocktail that causes the face to lose its natural fat and collagen, making it look sunken. It’s been called the scarlet letter of HIV, acting as a magnet for stigma.
Bio-Alcamid was supposed to fix all that. It’s a simple enough product – a gel that’s injected directly into the affected parts of the face. Polymekon, the Italian producer, billed it as a permanent, cost-effective, safe way to tackle the condition. It even came with an instructional DVD.
It seemed too good to be true.
And maybe it was.
The problems
Not long after it was approved for sale in Canada, in 2006, problems started cropping up. Israel, Scotland, Mexico and the Netherlands all began reporting problems with Bio-Alcamid: infections, inflammation and migration (when the implant would move throughout the face). It never even received approval in the United States.
The Canadian government, on the other hand, gave the green light after only a short-term, 96-week study.
But follow-up studies showed that between 19 and 25 percent of users contracted infections. Another 25 percent had other, less severe, complications with the product, according to one study. There exists a stack of literature released in recent years that shows the high complication rate that starts around four years after the first injection of Bio-Alcamid.
Pur Medical Corporation, a Toronto-based company established to distribute Bio-Alcamid, commissioned the original trials and distributed the product for three years.
“Would I do things differently?” says former Pur president John McCahill. “I don’t know.”
McCahill says they recognized some of the complications early on, and, working with Toronto’s Maple Leaf Medical Clinic, they issued revised instructions and guidelines to help doctors avoid causing complications. He says infections were usually caused by dental work, which introduced foreign bacteria to Bio-Alcamid. Even with the new warnings, complications continued.
Pur got out of the business of distributing Bio-Alcamid in 2007. The company became inactive after that.
Mona Loutfy, an infectious disease specialist who focuses on HIV/AIDS, oversaw the 2004 clinical trial for Maple Leaf and has co-authored several follow-up studies that show the high rate of complications associated with Bio-Alcamid.
She confirms McCahill’s theories – that any work done that could expose the Bio-Alcamid capsules to foreign bacteria could have caused the infections. Yet, Bio-Alcamid was designed to develop a protective capsule around itself (to prevent infection), but that protection appears to have failed.
After consensus formed in the medical community that the risks associated with Bio-Alcamid outweighed its benefits, it was taken off the shelves everywhere in Canada, with the last batch sold in 2007.
The reaction
Despite its faults, many still swear by Bio-Alcamid.
Bradford McIntyre is a blogger who has been living with HIV since 1984. McCahill contacted him eight years ago to participate in the clinical trial of Bio-Alcamid.
“It changed my life,” McIntyre says. “People have stopped looking at me like I’m sick.”
Loutfy points to cases like McIntyre’s to show that, while there have been problems, Bio-Alcamid is still effective at treating lipoatrophy.
“They were asking desperately for something,” she says. “At the time, there was nothing else available.”
In her study, 75 percent of participants said they are still satisfied with the injection. While today there are alternatives – drugs like Sculptra, which helps rejuvenate the collagen in patients’ faces and can combat moderate lipoatrophy – most alternatives at the time were costly or unavailable in Canada. The need for a new drug is now dwindling, as the antiretroviral cocktail has improved and facial wasting has become less of a problem.
Still, Loutfy says, “it would have been nice to know the side effects.”
Others agree.
Many took to the internet to share their stories of Bio-Alcamid. Plastic-surgery message boards are awash with horror stories.
It all culminated in a class-action lawsuit that was filed in 2011. Bruce Lemer was the lawyer who took on the case.
Approached by an HIV-positive Vancouverite in 2011, Lemer now has “a volume of research” that gets “worse and worse” with each page, he says.
“I’ve seldom seen such black-and-white statements,” he says.
One plastic surgeon took to his website to express that – contrary to the advertising around the product – Bio-Alcamid is not always easily removed.
Despite a clear case, the class action fizzled after Lemer realized there was nobody to hold responsible. The distributors, including Pur Medical Corporation, were small fish and little more than middlemen. Even if Lemer won the case against them, they simply would not have the financial resources to cover the damages.
Polymekon, who held the insurance policy on Bio-Alcamid, benefited from standing behind the thorny thicket of Italian corporate law. Lemer says that going after them would have proved immensely difficult.
Bio-Alcamid’s dissatisfied hosts were left holding the bag.
Some were lucky, however. Any patients who approached Pur or Maple Leaf got help paying for Bio-Alcamid’s removal and medical care, say McCahill and Loutfy.
It was better than “beating myself up,” McCahill says.
Despite all this noise about Bio-Alcamid, it seems the Canadian government never caught on. Lemer filed an access-to-information request with Health Canada to learn whether problems with the filler made its way to the government. According to the documents he received, it didn’t.
The government uses a newsletter to publish notices of complications with various drugs and medical products. For Bio-Alcamid, Health Canada published only one notice of an adverse reaction, which led to a slight change to the product’s packaging. No investigation was ever conducted. Lemer says there is a disconnect between the multitude of problems and the government’s lack of response.
Health Canada has confirmed to Xtra that, normally, the producer of a drug is legally responsible to convey reports of adverse reactions, which – depending on their frequency and severity – can lead to an investigation. Doctors and patients, on the other hand, are merely encouraged to report problems.
It’s unclear in this situation, since the producer isn’t Canadian, whether Pur Medical was required to report the adverse reactions it received, of which there were many.
The Italian company was let off the hook.
The company
Polymekon started as a family affair. Two brothers – Carmelo, the scientist and Biagio, the businessman – who were responsible for bringing Bio-Alcamid worldwide ran it.
Xtra contacted Carmelo Protopapa to ask about his experience with Bio-Alcamid. He, too, highlighted the great work that can be done with Bio-Alcamid.
“My wife has it, and mother has it,” Protopapa says. “They don’t have any problems.”
But, it seems, he was also one of the first to recognize the problems associated with the product.
Protopapa filed a lawsuit against Polymekon in 2005, after issuing several warnings about Bio-Alcamid that were ultimately ignored. “I filed legal proceedings to stop everything,” he says. “I lost.”
Protopapa spent months lobbying his brother, the CEO, to issue a warning to doctors. Bio-Alcamid, he said, couldn’t form a protective capsule around itself if it were too spread out across the face – and that would leave it open to infection. The executives weren’t interested.
“They only think of the money,” Protopapa says.
Bio-Alcamid is still being used, especially in parts of Asia, and has almost no complications. That, says Protopapa, is because the risk was finally recognized and Bio-Alcamid is now being used only for small plastic surgery procedures. They should have recognized that in 2005, he says, rather than keeping quiet about the side effects.
He explained some of these problems to one Canadian plastic surgeon who worked extensively with Polymekon. “He explained to me that it wasn’t important to remove it, because [Bio-Alcamid] doesn’t have any complications,” he says. “That wasn’t a smart answer.”
Protopapa says Polymekon was more than ready to cover the cost of removing Bio-Alcamid, but only if it could be proved that its malfunctioning was due to a manufacturing error. That’s no easy task. That deal was struck with several AIDS groups, and it appears that no Canadian patients ever had recourse to take that option.
Protopapa says he regrets finding out about the problems so late.
“I should have stopped before,” he says.

 Why you can trust Xtra
Why you can trust Xtra


