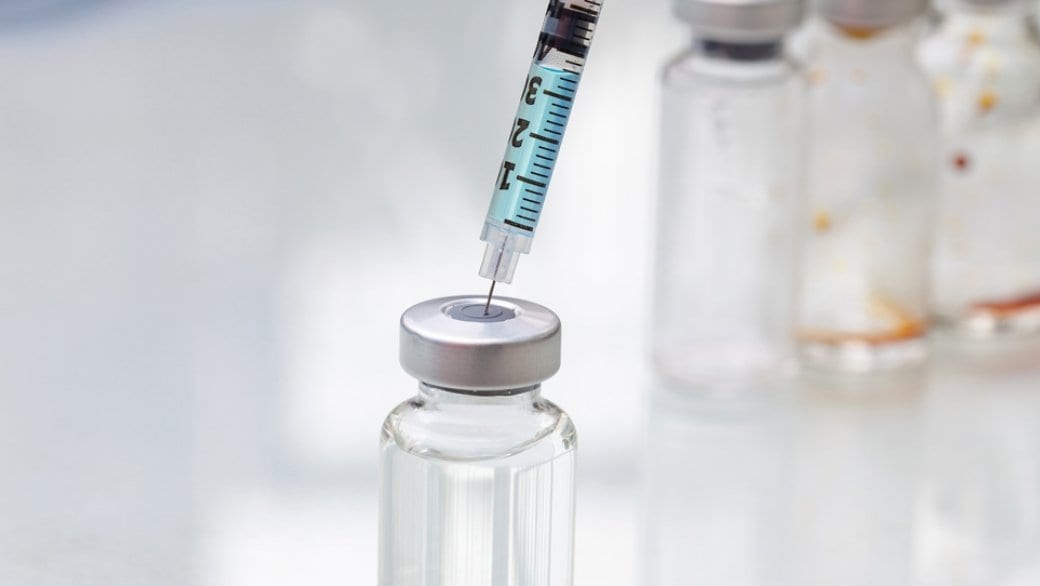
The first clinical trial for a long-acting injectable drug that could prevent the transmission of HIV began last month.
The purpose of injection is similar to using Truvada as PrEP, but instead of taking a pill daily, the anti-HIV drug, cabotegravir, is injected once every eight weeks with hopes that it will prevent the spread of HIV.
The study, called HPTN 083 sponsored by the National Institutes of Health, is to evaluate a revolutionary new injectable that could address the challenges some people may face around taking Truvada without fail every day.
I understand why it might be difficult for some people to adhere to a daily pill regimen, but for me it was never a problem. During the year and a half that I was on PrEP, I rarely forgot to take a pill. I set an alarm on my phone and scheduled it on my work and home computer calendar. Fortunately, my life was relatively routine so it was easy enough to remember.
Though even I slipped up occasionally. The first time that I forgot to take a pill was when I was on vacation in New York. I spent the night with someone I met at The Black Party and didn’t get back to my hotel until mid-afternoon the next day. My alarm had gone off on time that morning when I was with the guy, but by the time I got back to my room I forgot to take it.
I bought a pill box the very next day and I didn’t forget to take my Truvada much after that.
Although the Centers for Disease Control and Prevention (CDC) recommend that PrEP be taken daily — and I advocate that 100 percent — research shows that taking Truvada even four times a week is still 96 percent effective in preventing the transmission of HIV. When taken every day, Truvada is considered more than 99 percent effective.
Admittedly, some people may have trouble taking a pill daily, or even four times a week — a point that one AIDS organization seized upon to hammer Truvada.
“The bottom line is that people won’t adhere and take the pill,” Michael Weinstein claimed when he spoke to BuzzFeed in 2014. Weinstein is the president of the AIDS Healthcare Foundation (AHF) and has become quite infamous for his anti-PrEP stance. “That’s what studies have shown. If this catches on as a public health strategy, that means there are going to be people who will take Truvada irregularly and some will be infected, and some develop drug resistance.”
In 2015, AHF published a campaign in gay publications and magazines nationwide titled, “The War Against Prevention,” which was an attack on PrEP. POZ and AHF then had it out after POZ fact-checked the ad.
AHF’s ad had chosen to focus on one poll in particular which claimed that 95 percent of the members of The American Academy of HIV Medicine were concerned their patients wouldn’t take Truvada daily. POZ pointed out that — in the same survey — 79 percent of doctors polled said they were very likely to prescribe PrEP to someone with an HIV-positive partner, and 66 percent would prescribe it to men who have sex with men who are at high risk of contracting HIV.
Adherence is a concern for any drug, they also noted; not PrEP in particular.
AHF responded to POZ’s fact check by fact-checking POZ’s article. It quickly became a tit-for-tat.
Regardless of what side of the fence you’re on with PrEP, I’d hope that news of cabotegravir, if it proves successful, would settle any adherence concerns, and would add yet another tool to the arsenal in the fight against HIV.
“It is essential to develop multiple effective HIV prevention modalities so the most vulnerable populations have a choice of preventive options,” the study’s protocol chair, Raphael J Landovitz, told News Medical.
Having even more options that suit the habits of all people makes us that much stronger. Between condoms, treatment as prevention (TasP), PrEP and now potentially cabotegravir, we’re certainly getting somewhere.
And the proof is in the pudding. The number of new HIV diagnoses in the United States dropped nine percent from 2010 to 2014, which means that we’re doing something right.
The clinical trial for the injectable drug will be held in eight countries in the Americas, Asia and Africa and will enroll 4,500 men who have sex with men, and transgender women who have sex with men, who are at high risk of contracting HIV and who are 18 years or older. Unfortunately, the results aren’t expected in 2021 so disagreements about adherence will likely continue.
Sadly, I’m almost certain that even if the injectable is a success, Weinstein and others in the anti-PrEP camp will not be satisfied. They’ll likely continue to push back against such innovations, seemingly out of concern that they may dismantle condom culture.
I don’t want to lose condoms as an option either, but with 39,513 people diagnosed with HIV in the US in 2015 alone, it seems important to embrace all the tools we have available to us, condoms and new innovations alike. When new innovations come our way, why not keep an open mind and focus on their benefits rather than their deficits, in an effort to one day end this epidemic for good.
PrEP School runs every other Monday on Daily Xtra. Columnist Mike Miksche explores and navigates the world of sex and PrEP.
 Why you can trust Xtra
Why you can trust Xtra


